Image


The FDA clearance allows Francis Medical to begin broader commercial deployment of the minimally invasive technology across the United States after years of clinical development, investment fundraising, and patient trials.
The company announced that the Vanquish system is designed to treat localized prostate cancer using targeted sterile water vapor, commonly referred to as steam, to destroy cancerous tissue while limiting damage to surrounding healthy structures inside the prostate.
Healthcare specialists say the development could represent an important advancement for men diagnosed with localized prostate cancer, particularly patients seeking alternatives to surgery or radiation treatments that may carry significant long-term side effects.
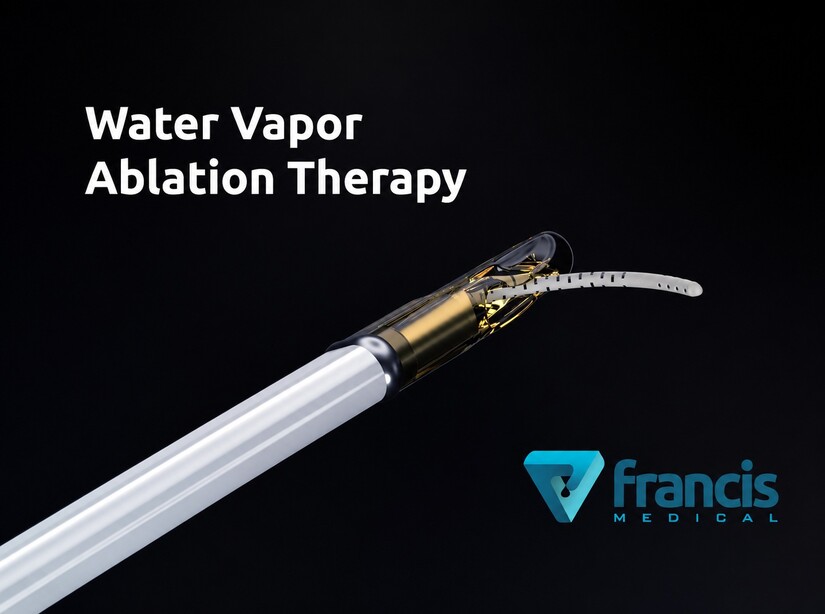
According to Francis Medical, the Vanquish Water Vapor Ablation System delivers targeted thermal energy directly into cancerous prostate tissue through sterile water vapor administered transurethrally, meaning through the urethra without external incisions.
The company says the thermal energy rapidly destroys cancer cells while minimizing collateral injury to nearby nerves and healthy tissue structures. The treatment is intended to reduce complications often associated with traditional prostate cancer interventions, including urinary incontinence and erectile dysfunction.
Prostate cancer remains one of the most commonly diagnosed cancers among men in the United States. According to the American Cancer Society, localized prostate cancer, meaning cancer confined to the prostate gland, accounts for the majority of new diagnoses.
Francis Medical says its technology specifically targets the approximately 84% of prostate cancer patients whose disease remains localized at the time of diagnosis.
Company officials describe the Vanquish procedure as a minimally invasive outpatient treatment intended to reduce hospital stays and recovery times.
According to Francis Medical:
The system is being positioned as an alternative between active surveillance and more aggressive interventions such as radical prostatectomy or radiation therapy.
Medical experts say many men diagnosed with localized prostate cancer struggle with treatment decisions because traditional therapies can carry lasting quality-of-life consequences.
Francis Medical was founded by medical device entrepreneur Michael Hoey, a longtime Minnesota healthcare innovator known for developing Rezūm therapy, another water vapor-based treatment used for benign prostatic hyperplasia, commonly known as enlarged prostate.
Hoey previously helped build Maple Grove-based NxThera before the company was acquired by Boston Scientific for approximately $406 million.
The founding of Francis Medical reflected an effort to apply similar water vapor technology toward cancer treatment rather than enlarged prostate conditions.
The company has since become part of Minnesota’s growing medical technology ecosystem, particularly in the northwest metro region, which has emerged as a major hub for healthcare device innovation.
Francis Medical expanded significantly during the past two years while preparing for regulatory approval.
In early 2025, the company secured approximately $80 million in Series C financing intended to support clinical trials, regulatory preparation, and commercialization efforts.
The funding round helped finance the company’s pivotal VAPOR 2 clinical study and scale manufacturing and operational infrastructure ahead of FDA review.
The company now plans to accelerate commercial deployment nationally following FDA clearance.
The FDA clearance followed data gathered through the company’s VAPOR 2 clinical study, which enrolled 235 men across 26 U.S. clinical sites.
According to company-released findings:
The prospective multicenter study focused primarily on men with intermediate-risk localized prostate cancer.
Participants in the study continue to be monitored over a five-year follow-up period to evaluate long-term cancer control and durability outcomes, with primary trial completion expected in April 2027.
Eligibility criteria for the study included:
Following FDA clearance, Francis Medical has already begun expanding clinical use of the Vanquish system at multiple healthcare centers across the country.
Participating medical centers currently include:
The company says the rollout is expected to expand further as physician training and implementation continue nationwide.
Although the initial FDA clearance focuses on localized prostate cancer treatment, Francis Medical executives have indicated the technology may eventually be adapted for additional cancer applications.
The company has publicly identified bladder cancer and kidney cancer as possible future targets for water vapor ablation research and development.
Healthcare analysts say successful expansion into additional oncology applications could significantly broaden the company’s long-term market potential.
The FDA clearance also highlights Minnesota’s continued influence in the global medical technology industry.
The Twin Cities region remains one of the nation’s largest medtech clusters, home to major healthcare innovators, research institutions, and medical manufacturing companies.
Maple Grove in particular has become increasingly recognized as a growing center for urology and minimally invasive medical device development.
Francis Medical’s headquarters is located at:
7351 Kirkwood Lane N, Suite 130, Maple Grove, Minnesota 55369.
As commercialization efforts begin, the company now enters a critical phase focused on physician adoption, insurance reimbursement pathways, long-term clinical outcomes, and market expansion.
For Minnesota’s healthcare and medtech sectors, the FDA clearance represents both a significant local business milestone and a potentially important development in the future treatment of prostate cancer.
MinneapoliMedia | Community. Culture. Civic Life.