Image


On a quiet afternoon in a suburban living room, a daughter asks her father a question so ordinary it should be effortless.
“Do you remember the lake house?”
She does not mean the deed or the address. She means the memory. The smell of sunscreen. The sound of a screen door closing. The dock that creaked underfoot. The laughter that once carried across water. She means the kind of recollection that proves a life was lived, not merely survived.
Her father looks toward the window as if the answer might be waiting outside. His face holds the faint polite smile of a man trying to meet expectations. Somewhere inside him, the image exists. But it cannot find the path back.
He pauses, then speaks quietly.
“I’m sorry,” he says. “I don’t.”
Alzheimer’s disease does not arrive like a storm. It arrives like erosion. It does not take a person all at once. It takes them in pieces. It begins with what feels like forgetfulness and ends as an unmaking. A slow reduction of a human being’s inner archive.
Families do not simply witness illness. They witness disappearance.
A parent forgets the name of a grandchild. A spouse repeats a question every five minutes, not because they want to, but because the brain cannot hold the answer. A neighbor who once knew every street in town becomes lost two blocks from home. The mind’s ability to store and retrieve life begins to fail.
This is the particular cruelty of Alzheimer’s. Many diseases threaten the body. Alzheimer’s threatens the continuity of self.
It is also one of the defining medical and social challenges of the twenty first century. More than 55 million people worldwide live with dementia today, and Alzheimer’s disease accounts for most cases. In the United States, more than 6.7 million Americans live with Alzheimer’s. By 2050, that number is projected to rise dramatically. Behind every number is a family improvising daily life around an unpredictable decline and a caregiving burden that can consume years.
Scientists have pursued treatments for more than a century. Drug developers have spent billions. Clinical trials have failed at a rate that would be intolerable in many other fields. Promising compounds collapse in human testing. Therapies that reduce plaques do not always produce meaningful cognitive improvement. Yet the need remains absolute because the scale of the crisis is expanding.
And this is why a new line of research has captured attention well beyond the world of neuroscience.
In 2025, researchers reported that engineered nanoparticles could dramatically reduce Alzheimer’s related pathology in mice, not by attacking brain cells directly, but by repairing the blood brain barrier and restoring a natural waste clearance system that the brain depends upon.
The claim is not that Alzheimer’s has been cured. The claim is something both more modest and, in its implications, more radical.
That a core component of the disease may involve the failure of the brain’s gatekeeping and cleanup infrastructure.
And that it may be repairable.
The human brain is not simply an organ. It is an environment. It functions because it maintains a fragile internal chemistry. Neurons exchange electrical impulses and chemical signals, and those exchanges are exquisitely sensitive to toxins, pathogens, and even ordinary fluctuations in blood composition.
Nature built a protective interface. It is called the blood brain barrier.
The blood brain barrier is not a wall. It is a living boundary formed by specialized endothelial cells lining the brain’s blood vessels, reinforced by tightly sealed junctions and supported by surrounding cells including pericytes and astrocytes. This barrier decides what enters the brain and what does not. Oxygen passes through. Glucose passes through. Certain carefully controlled nutrients and signaling molecules pass through. Many pathogens, toxins, and drugs do not.
But the barrier has another task that is easy to overlook. It removes waste.
The brain is metabolically intense. It produces byproducts of cellular activity, including proteins that must be cleared to prevent buildup. One of the most discussed of those proteins in Alzheimer’s research is amyloid beta.
Amyloid beta is a peptide produced through normal processing of a larger protein. Under healthy conditions, amyloid beta is produced and removed continuously. Problems arise when production outpaces clearance or when clearance mechanisms fail. Then amyloid beta accumulates. It can cluster and form plaques, which are among the best known pathological hallmarks observed in Alzheimer’s brains.
One of the key clearance mechanisms involves a receptor protein called LRP1, low density lipoprotein receptor related protein 1. LRP1 is expressed at the blood brain barrier and helps transport amyloid beta from the brain across the barrier into the bloodstream, where the body can eliminate it.
In plain terms, LRP1 is part of the brain’s garbage removal infrastructure.
If that infrastructure breaks down, waste accumulates.
If waste accumulates, the environment becomes toxic.
If the environment becomes toxic, neurons suffer.
This is not only a biochemical story. It is also, in a deeper sense, an infrastructure story.
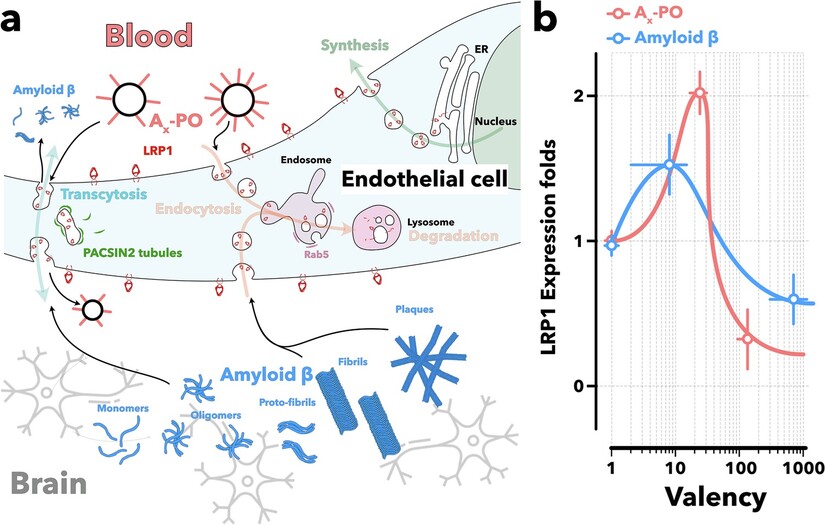
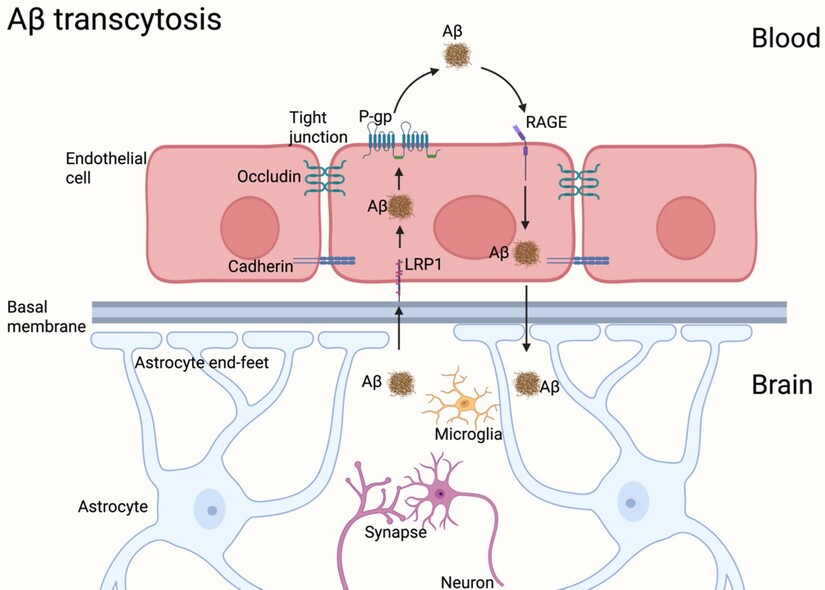
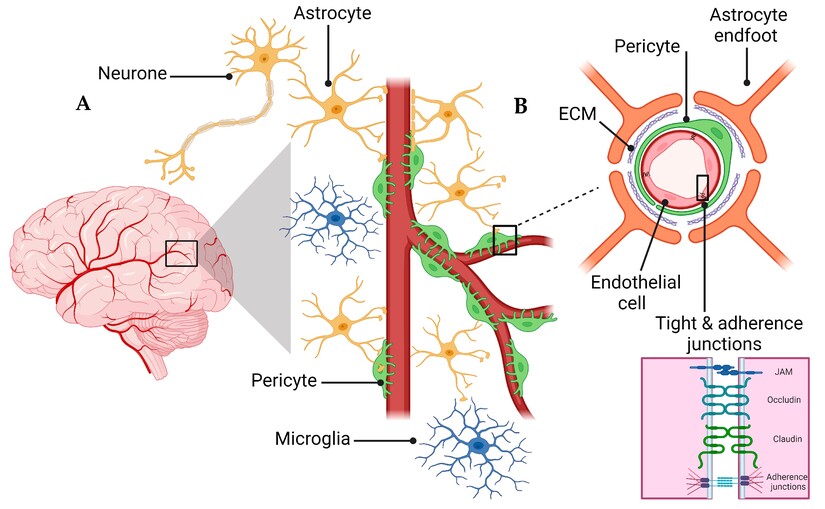
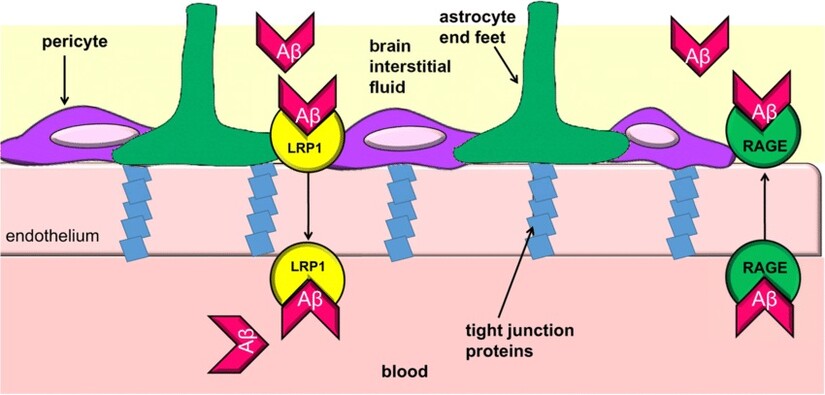
The blood brain barrier can be understood as the brain’s border control system and sanitation department combined. Nutrients enter through regulated transport. Waste leaves through regulated transport. LRP1 is one of the crucial exit gates.
In Alzheimer’s disease, the exit gates may narrow. The border may become leaky. And the result is an environment where harmful proteins are not removed efficiently, allowing pathological accumulation over time.
This is one reason researchers have begun shifting from a simplistic plaque centered view of Alzheimer’s toward a broader systems failure model that includes vascular integrity, barrier function, and immune response.
For decades Alzheimer’s research has been shaped by the amyloid cascade hypothesis, the idea that amyloid accumulation triggers downstream events that include inflammation, synaptic dysfunction, tau pathology, neuronal death, and cognitive decline.
The hypothesis has strong evidence. Genetic forms of Alzheimer’s often involve mutations that increase amyloid production or alter amyloid processing. Amyloid plaques are observable in many Alzheimer’s brains. Imaging technology can detect amyloid in living patients. The biology is not imaginary.
But the hypothesis also produced a painful reality. Even when researchers succeeded in reducing amyloid plaques, the clinical outcomes were often modest. Some drugs reduced plaques without producing meaningful improvement in cognition. Others produced side effects. The disease did not yield as easily as the plaque image suggested it might.
That led to deeper questions.
What if plaques are part of the pathology but not the full story?
What if the brain’s clearance system is a central battleground?
What if Alzheimer’s is not only a disease of buildup, but a disease of failure to remove?
This is where the blood brain barrier research becomes important. Many studies have shown blood brain barrier dysfunction in aging and neurodegenerative disease. Inflammation, vascular damage, diabetes, hypertension, and other systemic factors may erode barrier integrity. The barrier becomes less selective. It may allow harmful substances to enter the brain. It may also lose efficiency in transporting waste out.
A compromised barrier can therefore accelerate the very pathology the brain is attempting to avoid.
An editorial about Alzheimer’s must explain what is being lost.
Memory is not a single thing. It is a set of processes.
Working memory holds information briefly. Long term memory stores experiences. Episodic memory holds personal events. Semantic memory holds facts. Procedural memory holds skills like riding a bike.
At the cellular level, memory involves synapses, the junctions where neurons communicate. When we learn, synapses change. Connections strengthen. Patterns become easier to activate. The brain is constantly rewiring itself, refining pathways of recall.
Alzheimer’s disrupts this at multiple levels.
Amyloid beta oligomers, small soluble clusters, may interfere with synaptic function even before plaques form. Tau, another protein central to Alzheimer’s pathology, forms tangles inside neurons and disrupts the internal transport systems that neurons use to deliver nutrients and maintain structure. Inflammation accelerates damage. Microglia, the brain’s immune cells, may become chronically activated and contribute to a destructive cycle.
The result is synaptic loss, neuronal dysfunction, and eventually neuronal death.
When synapses vanish, memory is not merely inaccessible. It may be gone.
This is why families often describe Alzheimer’s as a kind of living bereavement. The person is physically present, but the continuity that once connected today to yesterday is fractured.
In this scientific landscape, the nanoparticle research is compelling because it takes a different approach.
Instead of focusing on neurons directly, the therapy focuses on the gate that regulates the brain’s environment. It attempts to restore the clearance system that should be removing amyloid beta naturally.
In the reported mouse studies, engineered supramolecular nanoparticles were designed to interact with the blood brain barrier and the LRP1 mediated transport pathway. The particles were engineered to bind amyloid beta and promote its clearance out of the brain.
The concept is not simply to attack plaques.
The concept is to restore a natural flow, to restart something that has stalled.
If Alzheimer’s involves a breakdown of clearance, then repairing clearance could change the trajectory.
This is conceptually similar to how modern medicine treats many systemic illnesses. Sometimes the solution is not to remove the final symptom, but to repair the underlying process that should prevent the symptom from accumulating.
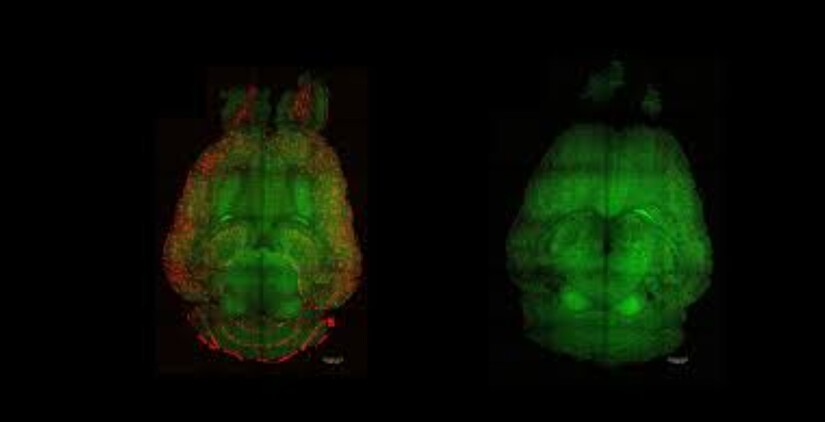
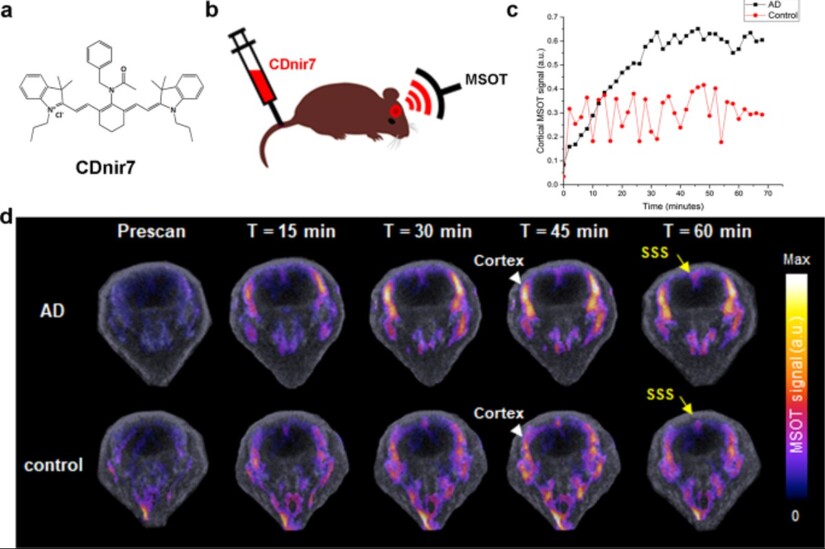
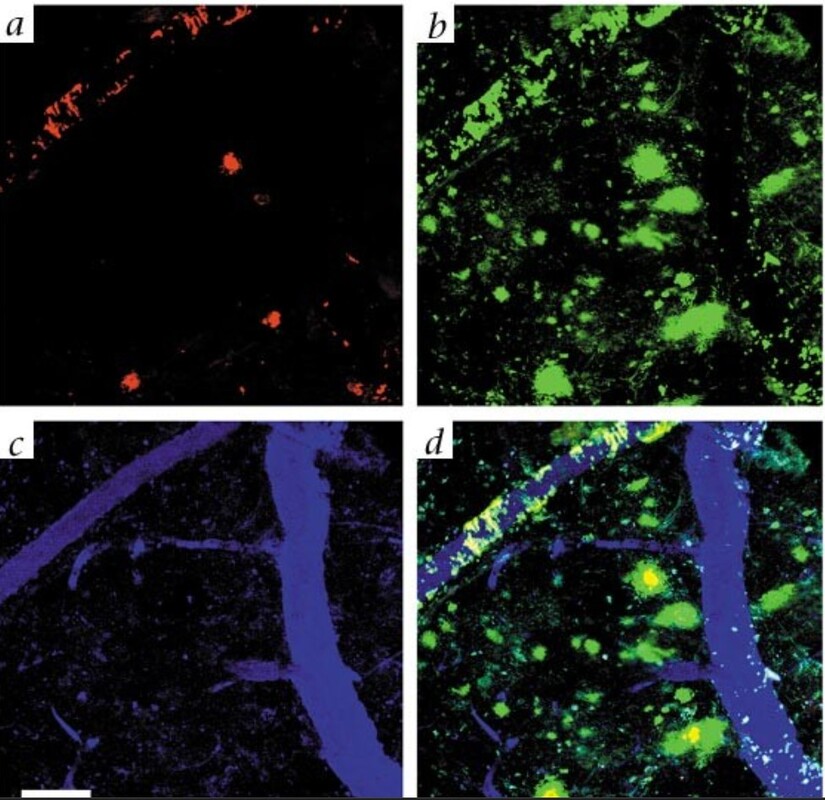
In imaging studies, amyloid plaques appear as bright clusters when tagged with fluorescent markers. The contrast between untreated and treated brain tissue can be visually startling. Dense plaque clusters in untreated animals can appear reduced after therapy, suggesting that clearance has increased and accumulation has diminished.
But this must be stated carefully. Plaque reduction in mice is not proof of clinical reversal in humans. It is evidence of a biological mechanism operating in an experimental model.
Still, in a field where progress is measured cautiously, a dramatic reduction in amyloid levels within a short time window is noteworthy.
In the reported experiments, multiple injections produced a large reduction of brain amyloid levels within about an hour in Alzheimer’s model mice. Older mice showed behavioral improvement months after treatment. Brain scans revealed fewer plaques.
These are not small claims.
They are also not human outcomes.
They are preclinical evidence.
It is easy to become cynical about Alzheimer’s research.
Families have been promised breakthroughs before. Headlines have declared cures that never arrived. For decades, Alzheimer’s has been the disease of nearly breakthroughs.
Yet preclinical work matters because it shapes the next generation of hypotheses. Even a therapy that does not reach the clinic can contribute to a deeper map of the disease.
The nanoparticle research reinforces an important shift.
Alzheimer’s is not only a brain cell problem. It is also a vascular problem. It is an infrastructure problem. It involves border control, clearance, and systems that maintain a healthy brain environment.
That shift opens new therapeutic strategies.
If the blood brain barrier is failing, can we repair it?
If clearance receptors like LRP1 are downregulated or malfunctioning, can we restore them?
If the barrier is leaky, can we tighten it?
This is the future frontier of Alzheimer’s research, the idea that neurodegeneration may be slowed by repairing the interface between blood and brain.
This is not the first time Alzheimer’s research has pivoted.
In 1906, Alois Alzheimer described plaques and tangles in the brain of a patient. Over decades, scientists identified amyloid beta and tau. In the late twentieth century, genetic discoveries strengthened amyloid centered models. In the early twenty first century, imaging and biomarkers revolutionized diagnosis, enabling researchers to detect pathology earlier.
But the clinical outcomes remained limited.
The field now recognizes that timing matters. By the time symptoms appear, pathology has been developing for years or decades. Intervening late may be too late.
This is why therapies aimed at clearance systems are so intriguing. If you can restore clearance early, you might prevent buildup. If you can prevent buildup, you might prevent downstream damage.
This is not guaranteed.
But it is a coherent strategic shift.
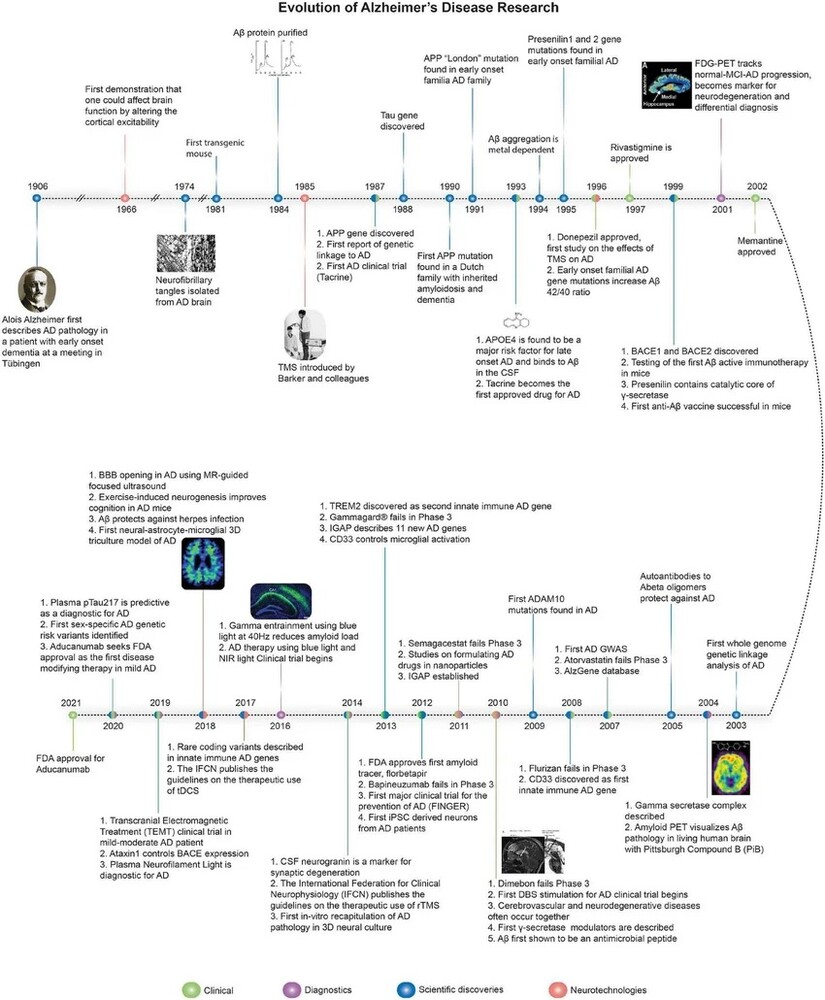
A century of research tells a story of persistence, failure, and gradual insight.
1906: Alois Alzheimer documents plaques and tangles.
1970s and 1980s: Amyloid beta is characterized and linked to plaques.
1990s: Genetic mutations tied to familial Alzheimer’s strengthen amyloid centered frameworks.
2000s and 2010s: Repeated clinical trial failures force reconsideration of disease complexity.
2010s: Imaging and biomarkers allow earlier detection and staging.
2020s: Antibody therapies modestly slow decline in some patients, while concerns remain about side effects, cost, and access.
2025: Nanoparticle research proposes a new intervention point, the blood brain barrier clearance system.
The timeline is not a victory march. It is a record of how difficult it is to treat a disease that is not one disease but a convergence of processes tied to aging.
Alzheimer’s is not only a biomedical problem. It is a demographic inevitability unless science changes the curve.
Human beings are living longer than at any point in recorded
history. This is one of the great achievements of modern civilization. But longevity changes the disease profile of society. As infectious disease declines in many regions, chronic conditions of aging rise.
Age is the greatest risk factor for Alzheimer’s.
Global projections suggest the number of people living with dementia could rise from about 55 million today to more than 139 million by 2050.
This growth will not be evenly distributed. High income nations already carry a large burden because populations are older. But the fastest growth is expected in regions where life expectancy is rising quickly.
This is crucial because it means Alzheimer’s will increasingly challenge healthcare systems that are still building foundational infrastructure. Countries that have not fully built long term care capacity will face dementia without an adequate caregiving scaffold.
The economic burden is already immense. Global dementia costs exceed one trillion dollars annually. But money is not the full measure. Alzheimer’s reorganizes family structures. It strains social safety nets. It creates a hidden workforce of caregivers, often unpaid, often exhausted, often invisible.
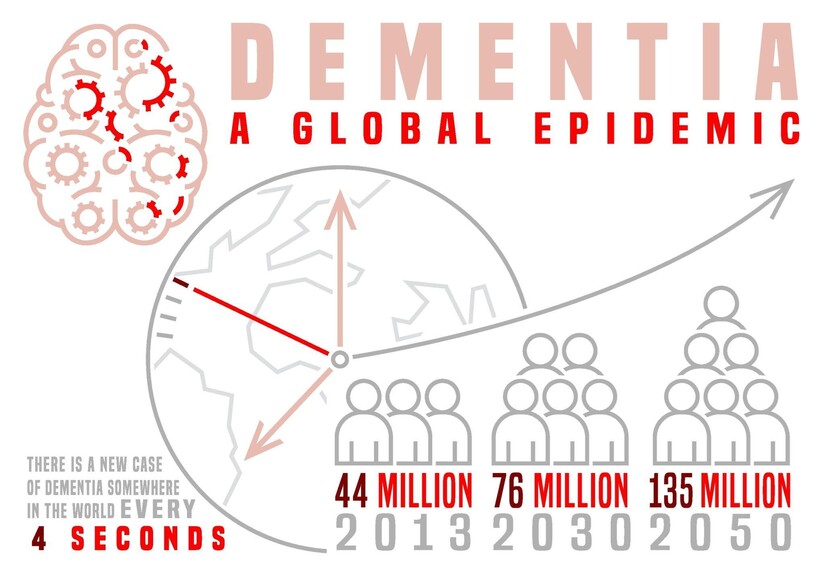
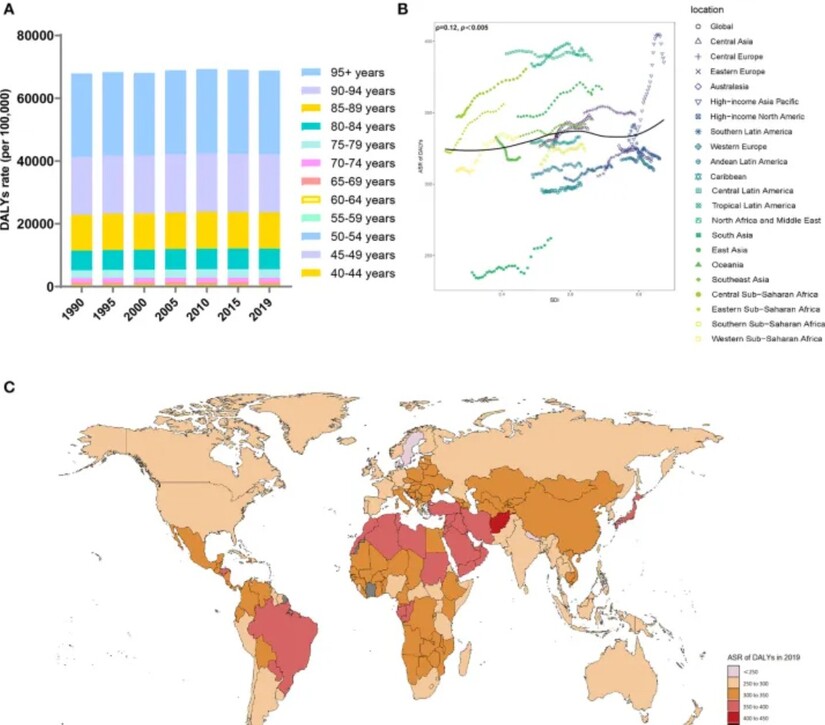
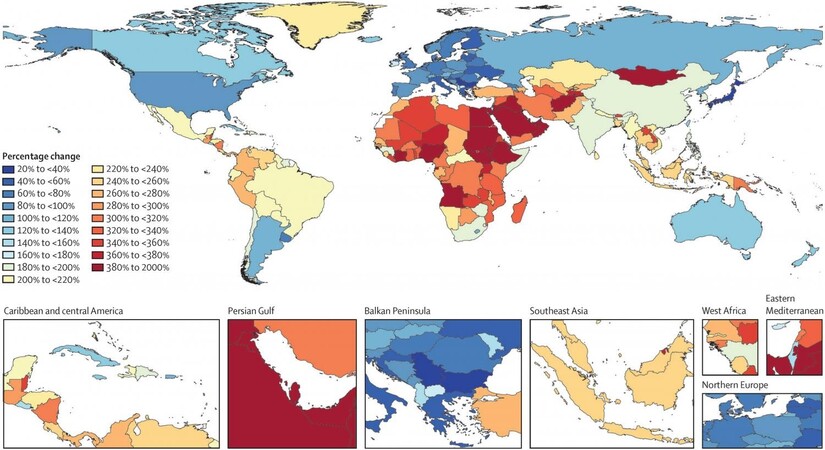
A global map of projected dementia prevalence is not simply a graphic. It is a warning.
It shows that Alzheimer’s will not remain confined to certain countries or demographics. It will expand with aging populations. It will become one of the defining health challenges of the century.
That is why even early stage research deserves attention.
Because the scale of need is accelerating faster than systems can adapt.
In the United States, the costs of Alzheimer’s care are often measured in billions. But those numbers can obscure what families actually experience.
The first stage of the disease often involves missed bills, confusion about medications, and unsafe driving. The middle stage can involve wandering, aggression, paranoia, and deepening dependence. The late stage often involves full time care, inability to communicate, and the need for feeding and bathing support.
Families make choices that reshape their lives. Adult children reduce working hours or leave jobs. Spouses become caregivers into their own old age. Savings are drained. Long term care facilities become necessary. Then the difficult question becomes financial. Who can afford care? Who has insurance? Who qualifies for public support? Who falls into the gap?
This is how Alzheimer’s becomes not only a medical crisis but a social justice issue.
If a breakthrough arrives, access will matter.
If treatments are expensive, they may widen inequality in who gets to preserve memory and who does not.
There is also a deeper ethical layer.
If science develops therapies that preserve cognitive function, society will face new questions.
How early should screening occur?
Who should receive preventive treatment?
How do we balance risk and benefit when intervening in people who have no symptoms yet?
How do we ensure that memory preservation does not become a luxury good?
Alzheimer’s therapies also intersect with questions of identity. Memory is not a simple commodity. It is the foundation of who a person is. Preserving it is not only a clinical goal. It is a moral and relational goal.
It is the difference between recognizing your child and not.
It is the difference between knowing your own name and losing it.
The nanoparticle research is not a cure.
It is a strategic hypothesis supported by dramatic preclinical results.
Its significance lies in what it suggests about Alzheimer’s biology.
That the blood brain barrier may not be a passive bystander.
That clearance may be as important as production.
That repairing infrastructure may matter as much as attacking symptoms.
This is a powerful shift because it invites the possibility of prevention.
If clearance systems can be restored early, pathology might be slowed before extensive neuronal loss occurs.
In Alzheimer’s, timing is everything.
Once neurons and synapses are lost, rebuilding memory becomes far more difficult.
So the future of Alzheimer’s treatment may depend not only on plaque removal, but on catching the disease earlier and maintaining the brain’s ability to protect and clean itself.
In the living room, the daughter does not correct her father. She does not force the memory. She holds his hand and sits with him in the quiet.
This is what families do. They become the external memory when internal memory fails. They become the map. They become the archive.
They remind. They repeat. They maintain routines that the brain cannot maintain on its own.
And they wait.
They wait for science to offer something more than comfort.
The nanoparticle research is a reminder that the frontier is still moving. That the brain’s gatekeeper can be studied. That its failure is not mysterious. That its repair might be possible.
No single study ends Alzheimer’s. But studies like this move the field toward a deeper understanding of what the disease truly is.
A breakdown of clearance.
A compromise of borders.
A slow poisoning of a delicate environment.
If we can repair the gatekeeper, we might protect the environment.
If we can protect the environment, we might protect memory.
And if we can protect memory, we might protect something close to the core of what it means to be human.